You’re cleaning out your garage, spot an old car battery in the corner, and someone casually says, “Just toss it in water, it’ll be fine.” Sounds simple—but that’s actually one of the worst things you can do.
I’ve had people bring in batteries after trying things like this, not realizing how dangerous it can get. So the real question is: what happens if you throw a car battery in water?
A car battery isn’t just a sealed box—it’s packed with acid, lead plates, and stored electrical energy. When water gets involved, especially the wrong kind or in the wrong situation, it can lead to short circuits, chemical reactions, leakage, and even serious safety risks.
This isn’t just about curiosity—it’s about avoiding costly damage, environmental harm, and potential injury. I’ve seen batteries completely ruined, tools damaged, and in some cases, people putting themselves at risk just because they didn’t understand what’s inside that heavy box.
I’ll break down exactly what really happens, the risks most people ignore, and what you should do instead. You’ll get clear, practical advice so you don’t end up turning a simple mistake into an expensive—or dangerous—problem.

Image by SafeAndLegalThrills
The Chemistry of Immersion: What Actually Happens?
To understand the reaction, we first need to look at what is inside the battery. Depending on the type of battery, the interaction with water changes slightly, but the outcome is always hazardous.
What It Is: Lead-Acid and Lithium-Ion Composition
Lead-Acid (Flooded, AGM, and Gel): These batteries contain lead plates submerged in a liquid or gelled electrolyte solution of sulfuric acid H_2SO_4 and water H_2O.
Lithium-Ion (LiFePO4 and NMC): These batteries contain highly reactive metals, organic carbonate solvents, and lithium salts.
How It Works: The Reaction with Water
When a flooded lead-acid battery is submerged in water, the concentrated sulfuric acid escapes the casing through the vent caps or a cracked housing. When sulfuric acid mixes with water, it creates an exothermic reaction—meaning it releases a massive amount of heat.
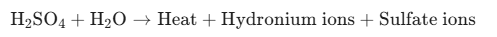
In addition to thermal danger, the reaction generates hydrogen gas H_2, which is highly flammable and can ignite upon contact with even a small spark. If the battery has a charge remaining, short-circuiting the terminals through water creates electrolysis, rapidly separating water into hydrogen and oxygen while corroding the lead plates and leaching heavy metals.
When and Why This Matters
Flooding and Natural Disasters: If your vehicle or off-grid battery bank is submerged during a flood, these reactions happen inside the equipment, posing an explosion and toxicity risk.
Accidental Drops: Dropping a marine battery overboard or an ATV battery into a creek can instantly poison the local ecosystem.
Real-World Insights and Tips
The Acid Spill Myth: Some people believe that diluting sulfuric acid with water neutralizes it. While it dilutes the concentration, it creates a larger volume of acidic water that spreads rapidly, killing aquatic life and stripping protective coatings off metal surfaces.
Comparing Battery Technologies: Risks and Performance
Not all batteries react to the environment in the same way. Below is a detailed comparison of common energy storage systems found in vehicles, solar arrays, and UPS backups.
| Battery Type | Nominal Voltage | Lifespan | Primary Use Case | Water Hazard Level | Pros | Cons |
| Flooded Lead-Acid | 12V (2.1V/cell) | 3–5 Years | Automotive, RVs, Golf Carts | Extreme (Acid + Lead contamination) | Low cost, high surge current | Heavy, requires watering, acid spill risk |
| AGM (Lead-Acid) | 12V | 4–7 Years | Motorcycles, Powersports | High (Heavy metal leaching) | Maintenance-free, mountable | Sensitive to overcharging |
| Gel (Lead-Acid) | 12V | 5–8 Years | Marine, Deep-Cycle | Moderate-High (Lead contamination) | Deep discharge recovery, no spills | Slower recharge times |
| LiFePO4 | 12.8V / 3.2V cell | 10+ Years | Solar, RV, Off-Grid | Critical (Thermal runaway hazard) | Lightweight, high efficiency | High upfront cost |
Battery Types and Their Practical Maintenance
1. Flooded Lead-Acid Batteries
- What it is: The traditional battery type featuring removable caps to check the electrolyte levels.
- How it works: Uses a liquid mixture of sulfuric acid and water. As it charges, it produces hydrogen and oxygen gases through gassing.
- When and why it should be used: Excellent for starting internal combustion engines where high burst currents are required, as well as budget-friendly off-grid storage.
- Real-World Tips: Always check water levels using distilled water. Never top up with tap water, as minerals will cause internal short circuits.
2. Lithium Iron Phosphate LiFePO_4
- What it is: The gold standard for modern deep-cycle and off-grid solar storage.
- How it works: Utilizes lithium salts as the electrolyte and a stable iron-phosphate cathode.
- When and why it should be used: Replaces lead-acid batteries when you need a lighter, faster-charging, and longer-lasting system with a built-in Battery Management System (BMS).
- Real-World Tips: Do not charge below freezing ($0^\circ\text{C}$ or $32^\circ\text{F}$) unless the battery has a low-temperature charging cutoff or an internal heating pad.
Step-by-Step Guide: Safely Testing and Charging Your Battery
To avoid needing to dispose of your battery prematurely or risking a thermal event, proper maintenance and diagnostic routines are essential.
Step 1: Safety First
Before interacting with any battery, put on protective safety glasses, acid-resistant gloves, and remove all conductive jewelry.
Step 2: Visual Inspection
Check the outer casing for bulging, cracks, or crystalline white or blue-green corrosion on the terminals. If the casing is deformed, the battery must be replaced immediately.
Step 3: Voltage Testing Using a Multimeter
- Set your multimeter: Turn the dial to the DC Voltage setting.
- Measure the voltage: Place the red probe on the positive terminal and the black probe on the negative terminal.
- Fully charged 12V lead-acid battery: 12.6V to 12.8V.
- Discharged lead-acid battery: 11.8V or lower.
- Fully charged 12V LiFePO4 battery: 13.2V to 13.6V.
Step 4: Connecting the Charger
- Match the chemistry of the charger to the battery. Using a lead-acid charger on a lithium battery can bypass the BMS and cause a fire.
- Set the charging current to roughly 10% to 20% of the battery’s total amp-hour (Ah) capacity to prevent overheating.
Real-World Usage Scenarios and Common Mistakes
1. Cars and Motorcycles
Scenario: Leaving your vehicle parked at the airport for a month, leading to a deeply discharged or dead battery.
Mistake: Attempting to jump-start a dead battery with an alternator, which can overheat the alternator and stress the battery.
Solution: Use a smart maintainer or a low-amp trickle charger to safely bring the battery back to optimal voltage.
2. Solar Systems
- Scenario: Off-grid solar arrays during a rainy or cloudy week.
- Mistake: Discharging a lead-acid battery bank below 50% depth of discharge, which causes rapid sulfation and permanent capacity loss.
- Solution: Upgrade to an LiFePO_4 bank to allow for deeper cycling without degrading the lifespan.
3. UPS and Backup Systems
- Scenario: Maintaining power for a home office or sensitive server rack during a storm.
- Mistake: Storing backup batteries in a hot garage or attic, where extreme heat accelerates the degradation of the electrolyte.
- Solution: Store backup units in a temperature-controlled environment between 15 degree Celsius and 25 degree Celsius.
Conclusion and Expert Takeaway
Properly understanding the lifecycle, maintenance, and safe disposal of your batteries is about more than just avoiding legal fines—it protects your equipment, your property, and the environment. Tossing a car battery into water leads to dangerous chemical reactions, toxic contamination, and explosive risks.
By familiarizing yourself with different battery chemistries, utilizing the correct chargers, and adhering to strict recycling guidelines, you can significantly extend the life of your power systems.
Pro-Level Tip: When reviving a deeply discharged lead-acid battery, always apply an initial desulfation charge at a very low current 0.5A to 1A for several hours before ramping up the amperage. This breaks down the lead sulfate crystals without warping the lead plates.
Frequently Asked Questions (FAQs)
Can a submerged car battery still explode?
Yes. When a battery is submerged, especially in salt water or moving water, short circuits occur internally and externally. This can cause the water inside the battery to undergo electrolysis, producing hydrogen gas that can ignite from a short-circuit spark.
What should I do if my car battery gets flooded in a storm?
Disconnect the negative terminal as soon as it is safe to do so. Remove the battery from the vehicle, inspect it for structural integrity, and have it tested at a local auto parts store. Never attempt to recharge a severely flooded battery.
How do I legally and safely dispose of an old car battery?
Bring the battery to an auto parts store, a local scrap metal yard, or a municipal hazardous waste collection center. Most retailers offer a core deposit refund or a recycling incentive when you drop off your old battery.
What happens if battery acid touches human skin?
Sulfuric acid causes rapid chemical burns. Immediately rinse the affected skin with plenty of cold water for at least 15 minutes, remove contaminated clothing, and seek medical attention if the burning persists.


